We focus on quality, flexibility & expertise.
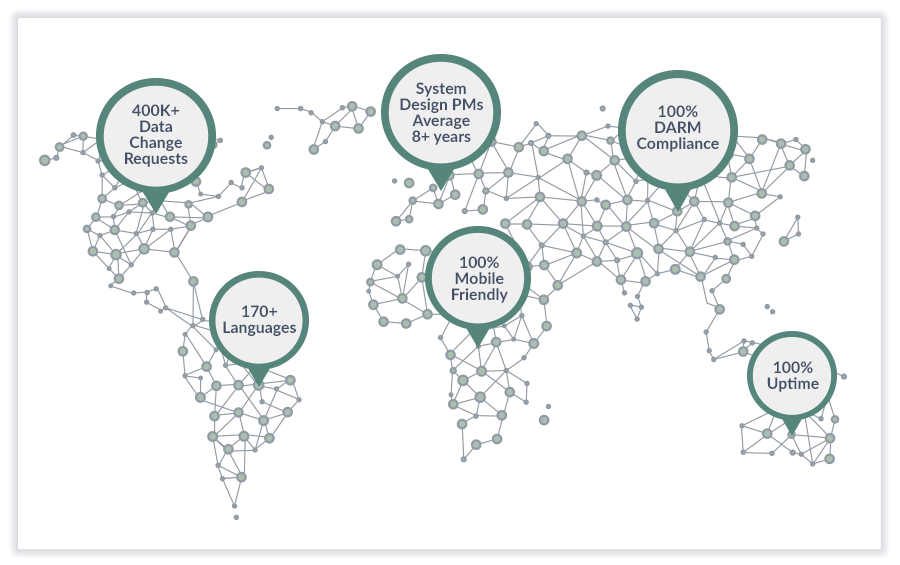
Cenduit, an IQVIA business, is a recognized industry leader in the IRT market and eClinical field, and have implemented over 1,600 clinical trials requiring patient randomization, clinical supply distribution and returns/destruction accountability. In over 10 years of operation, Cenduit has worked closely with more than 100 global biopharmaceutical companies and has deep integrations with many CROs, across all geographies. We have successful ongoing relationships with leading clinical supply packaging and distribution organizations, as well as extensive integrations with eClinical systems such as EDC, CTMS and Labs. We are IRT focused - dedicated to quality-first and patient outcomes. Our core expertise is strongly focused on randomization of patients and the management of clinical trials supplies.
We offer clinical supply chain intelligence & clinical operations knowledge management through IRT-driven solutions & services.
- Randomization
- Patient Engagement
- Drug Accountability and Traceability
- Drug Allocation
- Trial Supply Management
- Professional Services
- Integration
- Reporting and Forecasting
- 24 Hour Global Help Desk
Our Quality System maintains a sharp focus on the quality of IRT.
Our global Quality System promotes consistent, compliant delivery of our IRT-driven services and successful management of your clinical trial throughout requirements gathering, development, system testing, User Acceptance Testing, deployment and especially maintenance.
History Snapshot: Bridging the Pathway in IRT Solutions
-
Idea and Inception
Formed in May 2007, Cenduit originated as a joint venture between two leading clinical development and clinical supply chain organizations: IQVIA and Thermo Fisher Scientific. Both companies recognized the increasing demand for patient randomization and drug supply management services and seized the opportunity to create a specialized IRT solutions provider.
-
Our Evolution
Cenduit has evolved into a comprehensive, global, eClinical solutions provider, dedicated to innovating and accelerating clinical trials, while driving better patient-centric outcomes.
-
Our Expertise
With more than a decade of eClinical solution expertise, Cenduit has established itself as an industry leading IRT and e-Clinical solutions provider. Our team has a proven track record, and a state-of-the-art platform that enables faster set-up times, easy integration with other systems, and seamless IRT delivery.
-
Our Dedication To You
Cenduit’s focus on continuous improvement within our world-class, customer aligned service organization, provides the best possible experience for clinical trial subjects, and helps to mitigate risks in the clinical trials process.
Contact our team to find the right solutions for your trials.